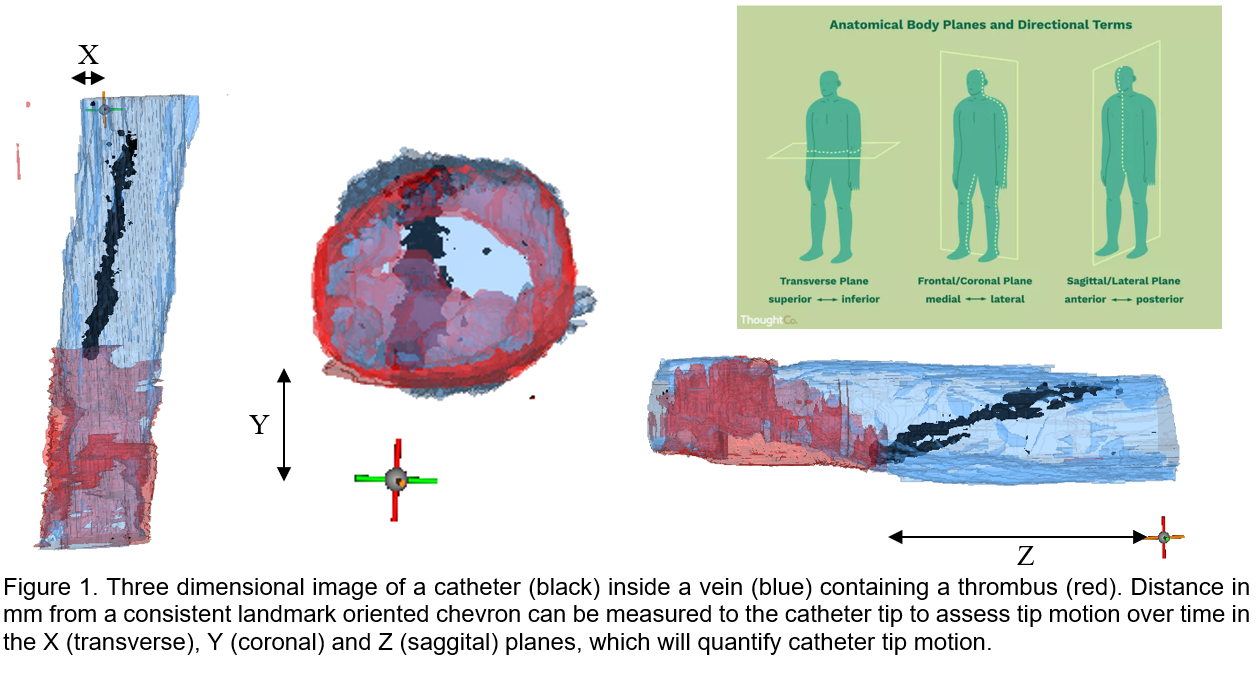
Impact of Catheter Stabilization on Catheter Micro-Motion and Function
The supplier BBraun has engineered a new type of peripheral intravenous catheter (PIVC) called the ‘Introcan Safety 3’ that contains a new type of stabilisation technology.
The AVATAR XLabs investigators would like to test this device against BBraun’s existing device (Introcan Safety) to determine if there is an improvement in device stability and whether this reduces blood clot formation and improves longevity of the device.
This study will be performed in human participants who meet the study screening criteria. We propose to use bilateral cannulation (i.e., cannulate both your left and right lower arms) and allow these PIVCs to stay in your arms for up to 72 hours to determine whether any advantage exists when using the different catheter types.
The position and motion of the two catheter types will be monitored using ultrasound, as well as standard observations to document whether the devices are working, and whether blood clots, local swelling, redness/pain, and device motion are observed.
Participants will also record their experiences with the devices to determine whether either catheter is associated with improved patient comfort.
Contact: Prof Andrew Bulmer, a.bulmer@griffith.edu.au
Trial registration link: https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=385245&showOriginal=true&isReview=true